
Isis May Hit Psoriasis Pay Dirt
Isis May Hit Psoriasis Pay Dirt
By Hal Plotkin
CNBC.com Silicon Valley Correspondent
Jun 26, 2001 12:00 PM
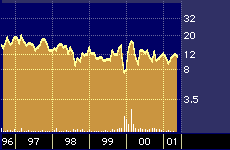
Isis Pharmaceuticals Inc. 5-year stock performance
Isis Pharmaceuticals Inc. {ISIP} could get more attention from investors after the firm’s weekend announcement that it will commence Phase II clinical testing of a new topical ointment for the treatment of psoriasis, an ailment that currently afflicts more than 7 million Americans. More than 150,000 new cases are reported each year.
Current psoriasis treatments are often found lacking, particularly in the most severe cases. Isis Pharmaceuticals is striving to develop a topical ointment-like treatment when most competitors, including Genentech {DNA} and Biogen {BGEN}, are developing treatments that require injections. That makes the stock particularly interesting.
“Isis Pharmaceuticals is going to be a real big winner,” says Berkeley, California-based Jim McCamant, who runs the American Health Care hedge fund and publishes the Medical Technology Stock Newsletter. “I think it could go to 30 [dollars a share].”
McCamant is a veteran biotech stock picker with one of the best track records in the industry. His model biotech portfolio, which was up 86 percent last year, has increased 746 percent since he initiated coverage of biotech stocks in 1987. McCamant’s aggressive biotech portfolio was up 53 percent last year, bringing total returns for that group of stocks to 2,474 percent since 1987.
“I think people are going to be hearing more about this over the week,” McCamant says, referring to the Isis announcement, which was made at the San Francisco meeting of the International Psoriasis Symposium, which concluded on Sunday.
“Everyone will prefer a topical to an injectable, and the previous experience with 2302 [a chemically similar Isis compound] means it’s pretty likely to be useful,” McCamant says.
ISIS Pharmaceuticals is a pioneer of a treatment approach called antisense that is also the foundation for pending Phase III trials of ISIS 2302 for the treatment of Crohn’s disease. The company’s antisense approach targets TNF-alpha, a naturally occurring compound that is implicated in the development and progression of many inflammatory, infectious and autoimmune diseases.
“The antisense dermatology platform has very broad potential,” Dr. F. Andrew Dorr, Isis’ vice president and chief medical officer, said in making the announcement. “We are using antisense to facilitate a better understanding of the molecular basis of skin diseases and can directly use that information to create antisense drugs in topical formulations.”
Isis’s stock is thinly watched by Wall Street standards, with just two firms, Needham & Company Inc. and Roth Capital Partners Inc. offering formal coverage, according to First Call Corp. Both firms have “buy” ratings on the stock.
Investors should, however, always be aware of the significant risks posed by investments in potentially volatile early stage biotech firms, even one that has had its drugs in development for as long as Isis Pharmaceuticals.
“Companies in the earlier stages of testing could be promising,” says Alfred Mansour, Ph.D., chairman and CEO of BiotechWatch.com, an online service based in Franklin, Michigan that tracks the progress of biotech firms.
But in general, Mansour says he sees the biotech testing process as something of an endurance contest, one that most new drugs don’t survive.
“You’ve got to think of it as an attrition problem,” he told CNBC.com earlier this year. “Maybe 50 percent of products in Phase I go on to the next stage of testing, then, maybe at best, less than 70 percent of the Phase II trials go on. The further you go the better your chances,” he says.
Assuming continued success, it will probably be at least 2003 before Isis will be in position to petition the FDA for approval to start selling the new psoriasis ointment. The stock is likely to be quite volatile between now and then, along with much of the often-gyrating biotech sector.
“I’d be a buyer of the stock right now,” says McCamant. “There’s going to be a lot of interest in Isis’s approach.”


